Pharmaceutical Packaging | Vibepedia
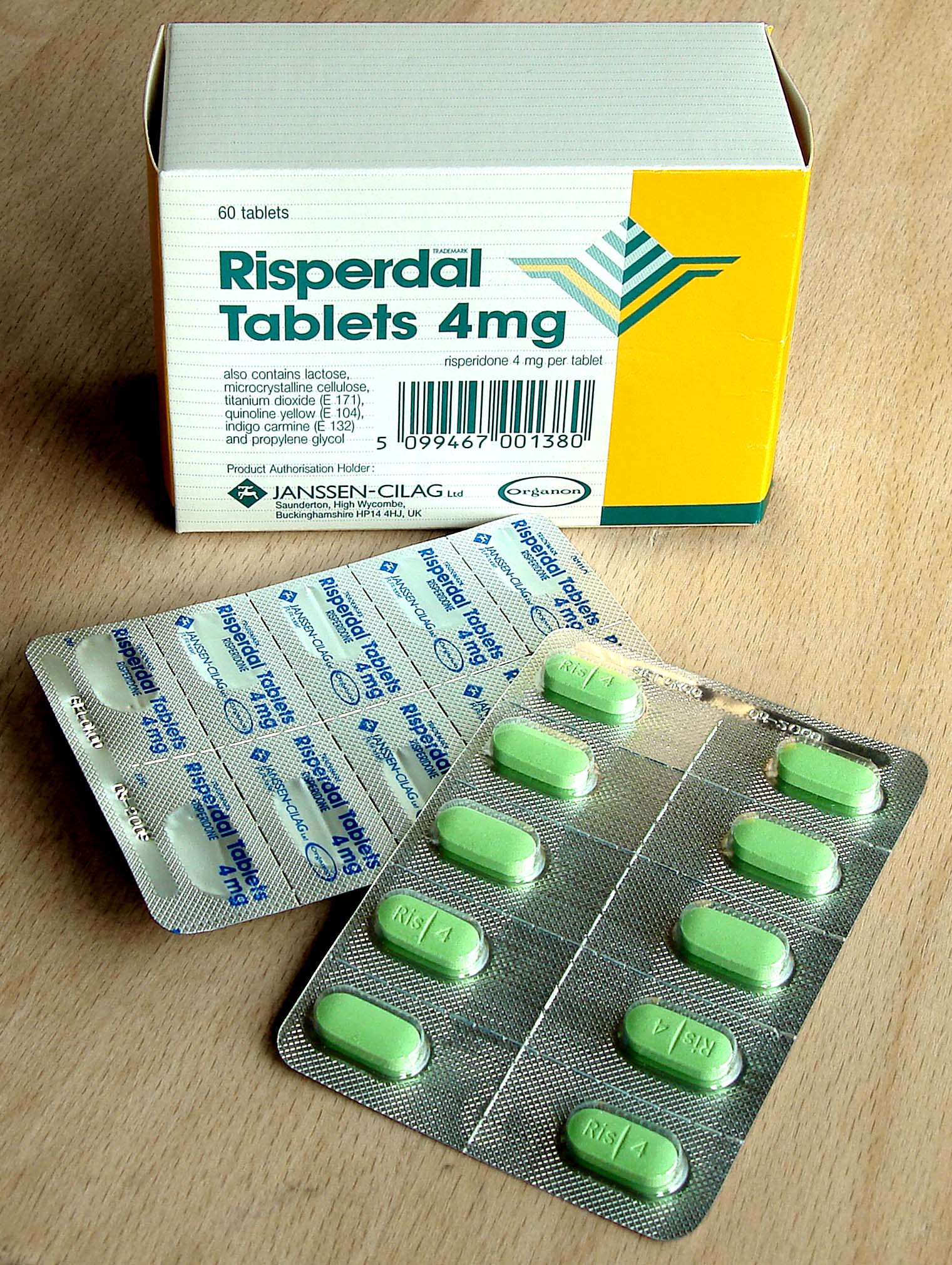
Pharmaceutical packaging is the intricate system of containers, closures, and labeling designed to protect medicinal products from environmental factors…
Contents
Overview
Pharmaceutical packaging is the intricate system of containers, closures, and labeling designed to protect medicinal products from environmental factors, ensure their integrity and efficacy throughout their shelf life, and provide essential information to healthcare professionals and patients. This highly regulated field balances the need for robust protection against moisture, light, oxygen, and microbial contamination with the demands of patient safety, tamper evidence, and ease of use. From the primary blister packs and vials that directly contact the drug to secondary cartons and tertiary shipping containers, every layer plays a vital role in maintaining product quality and preventing counterfeiting. The global pharmaceutical packaging market is driven by stringent regulatory requirements, the growth of biologics, and the increasing demand for patient-centric delivery systems.
🎵 Origins & History
The genesis of pharmaceutical packaging can be traced back to ancient times, where rudimentary containers like clay pots and leather pouches were used to store medicinal herbs and concoctions. The advent of glassblowing in the Roman era allowed for more sophisticated vials, offering better preservation. However, it wasn't until the 19th century, with the rise of mass production and the development of new materials like paperboard and early plastics, that pharmaceutical packaging began to resemble its modern form. Subsequent innovations underscored the growing emphasis on patient safety, a principle that continues to shape the industry today.
⚙️ How It Works
Pharmaceutical packaging operates through a multi-layered approach, each layer serving distinct protective and informational functions. The primary packaging, in direct contact with the drug, includes materials like glass vials for injectables, plastic bottles for oral solids, and aluminum-foil blister packs for tablets and capsules. These materials are chosen for their inertness, barrier properties against oxygen, moisture, and light, and their ability to maintain sterility. Secondary packaging, typically a carton, houses the primary container and often includes patient information leaflets (PILs) or prescribing information. Tertiary packaging, such as corrugated boxes, is used for bulk shipping and distribution, often incorporating tamper-evident seals and serialization codes to track the product through the supply chain, as mandated by regulations like the Drug Supply Chain Security Act (DSCSA).
📊 Key Facts & Numbers
The global pharmaceutical packaging market is a colossal enterprise. Within this market, plastic packaging accounts for a significant share, followed by glass and paperboard. The demand for sterile packaging is substantial. Regulatory bodies like the U.S. Food and Drug Administration (FDA) oversee packaging regulations, with a large percentage of all pharmaceutical products utilizing some form of plastic packaging for their primary containment.
👥 Key People & Organizations
Several key organizations and individuals have shaped the landscape of pharmaceutical packaging. The Pharmaceutical Packaging Consortium (PPC) plays a crucial role in setting industry standards and fostering collaboration. Companies like Amcor and Berry Global Group are titans in the manufacturing of pharmaceutical packaging solutions, supplying a vast array of bottles, films, and closures. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are paramount, dictating stringent requirements for safety, efficacy, and traceability. Innovators like Dow Chemical have also been instrumental in developing advanced polymer materials that enhance barrier properties and drug stability.
🌍 Cultural Impact & Influence
Pharmaceutical packaging's influence extends far beyond its functional role, deeply embedding itself into public perception and healthcare practices. The visual cues of packaging — the sterile white of a pill bottle, the reassuring blue of a blister pack, the clear instructions on a label — contribute to a patient's trust in a medication's efficacy and safety. Innovations like child-resistant caps have become ubiquitous, shaping parental habits and contributing to a significant reduction in accidental pediatric poisonings. Furthermore, the rise of serialization and track-and-trace technologies, driven by concerns over counterfeiting, has introduced a new layer of digital interaction, connecting the physical package to global supply chain databases and enhancing transparency for consumers and regulators alike.
⚡ Current State & Latest Developments
The current state of pharmaceutical packaging is characterized by rapid innovation and evolving regulatory pressures. The surge in biologics and complex drug formulations necessitates advanced packaging solutions offering precise temperature control and enhanced sterility, exemplified by the specialized cold-chain packaging developed for Pfizer-BioNTech's COVID-19 vaccine during the 2020-2021 pandemic. Sustainability is also a major driver, with manufacturers exploring biodegradable plastics and increased use of recycled materials, though balancing these with stringent regulatory demands remains a challenge. The implementation of serialization and track-and-trace systems, mandated by global regulations like the EU's Falsified Medicines Directive (FMD), is nearing full compliance in many regions, aiming to combat the growing threat of counterfeit drugs.
🤔 Controversies & Debates
Significant controversies and debates surround pharmaceutical packaging, primarily concerning material safety and environmental impact. The potential for extractables and leachables — chemical compounds migrating from the packaging into the drug product — is a persistent concern, leading to rigorous testing protocols and ongoing research into inert materials. For instance, debates continue regarding the safety of certain plasticizers like phthalates in medical devices and packaging. Furthermore, the environmental footprint of single-use plastics used in pharmaceutical packaging is a major point of contention, with calls for greater adoption of sustainable materials and recycling initiatives clashing with the absolute requirement for product integrity and patient safety. The cost implications of advanced, compliant packaging also spark debate, particularly for generic drug manufacturers and in emerging markets.
🔮 Future Outlook & Predictions
The future of pharmaceutical packaging points towards increased integration of smart technologies and a heightened focus on patient-centric design and sustainability. Expect to see more 'smart packaging' solutions incorporating sensors to monitor temperature, humidity, or even indicate if a dose has been taken, potentially linking via NFC or Bluetooth Low Energy (BLE) to patient apps. The drive for sustainability will likely lead to wider adoption of bio-based polymers and advanced recycling technologies, though regulatory hurdles will persist. Personalized medicine will also drive demand for smaller, more specialized packaging formats. Companies like Medtronic are already exploring connected packaging for drug delivery devices, hinting at a future where the package is an active participant in patient care, not just a passive container.
💡 Practical Applications
Pharmaceutical packaging finds critical application across the entire spectrum of medicine delivery. Primary packaging, such as glass vials and pre-filled syringes, is essential for injectable drugs, vaccines, and biologics, ensuring sterility and precise dosing. Blister packs, often made from PVC and aluminum foil, are ubiquitous for oral solid dosage forms like tablets and capsules, providing individual dose protection and tamper evidence. Bottles, typically made from HDPE or PET, are common for pills and liquids, often featuring child-resistant caps and induction seals for tamper evidence. Specialized packaging is also crucial for sensitive drugs requiring protection from light (e.g., amber glass bottles) or moisture (e.g., desiccant-lined containers). The development of advanced drug delivery systems, such as inhalers and transdermal patches, also relies heavily on specialized, functional packaging components.
Key Facts
- Category
- technology
- Type
- topic