Autophagy Research | Vibepedia
Autophagy, a fundamental cellular process meaning 'self-eating,' is the mechanism by which cells degrade and recycle damaged or unnecessary components…
Contents
Overview
Autophagy, a fundamental cellular process meaning 'self-eating,' is the mechanism by which cells degrade and recycle damaged or unnecessary components. Discovered as a survival strategy during starvation, its role has expanded to encompass cellular homeostasis, immune response, and the pathogenesis of numerous diseases. Research into autophagy, particularly macroautophagy, has exploded since the early 2000s, revealing its complex regulation and therapeutic potential in conditions ranging from neurodegenerative disorders like Alzheimer's disease to cancer and infectious diseases. The field is characterized by intense investigation into its molecular machinery, signaling pathways, and the development of pharmacological modulators, with Nobel Prizes recognizing key discoveries and a growing understanding of its dual role in health and disease.
🎵 Origins & History
The concept of autophagy, or 'self-eating,' traces its roots back to the 1950s with the pioneering work of Christian de Duve, who identified lysosomes as cellular organelles responsible for degradation. Early research, heavily influenced by studies on starvation and cellular survival, primarily viewed autophagy as a catabolic process for recycling cellular components during nutrient deprivation. The field remained relatively niche until the late 1990s and early 2000s, when significant advances in molecular biology and genetics, particularly the identification of autophagy-related genes (ATGs) in yeast by Yoshinori Ohsumi and others, propelled autophagy research into the mainstream, revealing its ubiquity and critical roles beyond mere starvation response.
⚙️ How It Works
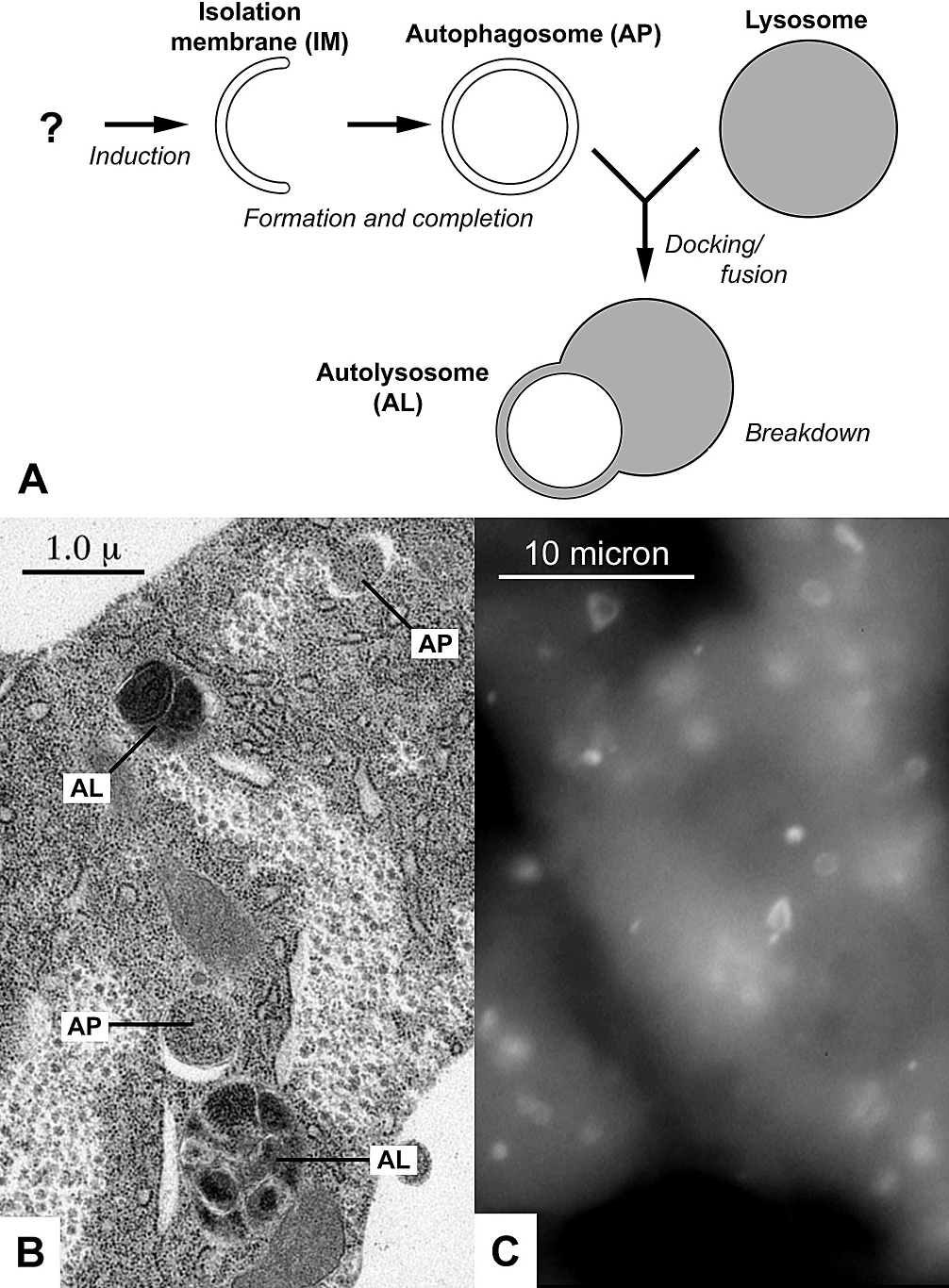
Autophagy operates through a complex, multi-step molecular cascade, primarily involving macroautophagy. The process begins with the induction of an isolation membrane, or phagophore, which expands and engulfs cytoplasmic cargo—ranging from damaged organelles like mitochondria to protein aggregates and invading pathogens. This double-membraned vesicle, now called an autophagosome, then fuses with a lysosome, forming an autolysosome. Inside the autolysosome, lysosomal hydrolases degrade the engulfed contents into basic building blocks like amino acids and fatty acids, which are then recycled back into the cytoplasm for cellular biosynthesis and energy production. Key molecular players include the ULK1 complex for initiation, the PI3K complex for phagophore formation, and ATG proteins like ATG5, ATG7, and LC3 (microtubule-associated protein 1A/1B-light chain 3) for elongation and cargo recognition. Different types of autophagy, such as chaperone-mediated autophagy (CMA) and microautophagy, involve distinct mechanisms for delivering substrates to the lysosome, highlighting the diverse strategies cells employ for internal cleanup.
📊 Key Facts & Numbers
Autophagy research has seen a dramatic surge, with the number of published articles increasing more than tenfold since the early 2000s. The field's significance was underscored by the 2016 Nobel Prize in Physiology or Medicine awarded to Yoshinori Ohsumi for his discoveries concerning the mechanisms of autophagy, recognizing over 5,000 citations for his foundational work. Global investment in autophagy-related drug development is projected to reach billions of dollars. Defects in autophagy have been implicated in over 70 human diseases, including various forms of cancer and neurodegeneration. It is reportedly estimated that 30-40% of cellular proteins are degraded via autophagy over a cell's lifetime.
👥 Key People & Organizations
The landscape of autophagy research is populated by a constellation of pioneering scientists and influential institutions. Yoshinori Ohsumi, a Nobel laureate, remains a central figure for his work on autophagy-related genes (ATGs) at the Tokyo Institute of Technology. David M. Sabatini at the Whitehead Institute made significant contributions to understanding mTOR signaling, a key regulator of autophagy, and later faced controversy regarding research misconduct allegations. Other key figures include Ralph Levine and Benjamin Neal, whose work at Yale University has elucidated novel regulatory pathways. Major research hubs include the National Institutes of Health (NIH) in the U.S., the Max Planck Society in Germany, and numerous university departments globally. Pharmaceutical companies like Gilead Sciences and Verastem Oncology are actively developing autophagy-modulating drugs, signaling a strong industry interest.
🌍 Cultural Impact & Influence
Autophagy research has permeated various scientific disciplines and captured public imagination, particularly through its connection to longevity and disease. The Nobel Prize awarded to Yoshinori Ohsumi in 2016 has reportedly boosted public awareness, framing autophagy as a critical cellular process for health maintenance. Its implications for aging have been widely discussed in popular science literature and media, linking autophagy modulation to potential anti-aging interventions. Furthermore, the dual role of autophagy—sometimes promoting cancer cell survival, other times acting as an anti-cancer mechanism—has generated considerable debate and media attention, highlighting the complexity of its biological functions. The visual representation of autophagosomes and autolysosomes has also become iconic in cell biology textbooks and scientific presentations, symbolizing cellular cleanup and renewal.
⚡ Current State & Latest Developments
The current frontier of autophagy research is characterized by a focus on therapeutic targeting and understanding its intricate regulatory networks. In 2024, significant progress is being made in developing selective autophagy modulators, moving beyond broad-spectrum inhibitors like chloroquine and hydroxychloroquine. Clinical trials are ongoing for autophagy inhibitors in various cancers, such as pancreatic and ovarian cancers, with companies like Gilead Sciences and Verastem Oncology leading the charge. Researchers are also exploring autophagy's role in neurodegenerative diseases, with efforts to enhance autophagic flux to clear protein aggregates in conditions like Parkinson's disease and Huntington's disease. The development of novel imaging techniques and biosensors allows for real-time monitoring of autophagy in living cells and organisms, providing unprecedented insights into its dynamic nature.
🤔 Controversies & Debates
A central controversy in autophagy research revolves around its paradoxical role in cancer. While autophagy can act as a tumor suppressor by clearing damaged components and preventing genomic instability, established tumors often hijack autophagy to survive chemotherapy, radiation, and nutrient deprivation, making them more resistant to treatment. This has led to debate on whether to inhibit or induce autophagy in cancer therapy. Another area of contention involves the precise mechanisms of autophagosome nucleation and expansion, with ongoing research refining models of how these membranes form and engulf cargo. Furthermore, the interpretation of autophagy markers, particularly LC3, has faced scrutiny, with calls for more rigorous validation to ensure accurate assessment of autophagic flux in different experimental contexts.
🔮 Future Outlook & Predictions
The future of autophagy research is poised for significant breakthroughs, particularly in precision medicine. Expect to see the development of highly selective autophagy modulators tailored to specific disease contexts and patient genotypes, moving beyond current broad-spectrum approaches. The integration of artificial intelligence and machine learning is accelerating the discovery of novel autophagy regulators and drug candidates, potentially identifying therapeutic targets that were previously overlooked. Research into the interplay between autophagy and the gut microbiome is expected to expand, revealing new avenues for treating metabolic and inflammatory diseases. Furthermore, advancements in in vivo imaging and single-cell analysis will provide deeper insights into how autophagy functions dynamically within complex tissues and during disease progression, paving the way for more effective therapeutic interventions by 2030.
💡 Practical Applications
Autophagy research has direct implications for a range of practical applications, most notably in the pharmaceutical industry. Drugs that modulate autophagy are being developed as potential treatments for cancer, aiming to sensitize tumo
Key Facts
- Category
- science
- Type
- topic